Abstract
Introduction: Acute myeloid leukemia (AML) is a heterogeneous disease with variable responses to therapy and clinical outcomes. Cytogenetics and molecular analyses help to stratify patients and to select therapy, especially regarding indication of hematopoietic stem cell transplant (HSCT) after achieving complete remission (CR). Patients in the intermediate cytogenetic risk category (~40%) represent a clinical dilemma regarding consolidation because of the high rate of relapse with chemotherapy alone and high rate of morbidity/mortality with HSCT. Consequently, we aimed to identify new prognostic markers to refine the risk stratification of this patients' subgroup and to identify which patients are most likely to benefit from HSCT.
Methods: We analyzed RNA sequencing data of 263 specimens from patients with de novo AML treated with curative intent including 165 patients with intermediate risk cytogenetics. Data from 24 586 genes were normalized as RPKM values with logarithmic transformation and standardization. Cox proportional hazard models were used to assess the prognostic impact of gene expression (GE) on overall survival (OS) and relapse-free survival (RFS). We performed univariate analyses (UVA) and multivariate analyses (MVA) adjusted for age and white blood cell count (WBC) at diagnosis, mutations in NPM1, FLT3, CEBPA, RUNX1, ASXL1, TP53 and DNMT3A and HSCT as a time-dependent (TD) covariate. Interaction between GE and TD-HSCT was tested in MVA for OS and RFS. Genes with a significant interaction between GE and TD-HSCT (p < 0.10) were retained for further analyses. GE of candidate markers were dichotomized using a bioinformatic method assessing hazard ratios (HR) and p values for all potential cut-offs to identify the most optimal threshold. The markers were finally reassessed as dichotomic variables for association with covariates, CR rates, RFS and OS. All statistical tests were two-sided with p values < 0.05 considered significant.
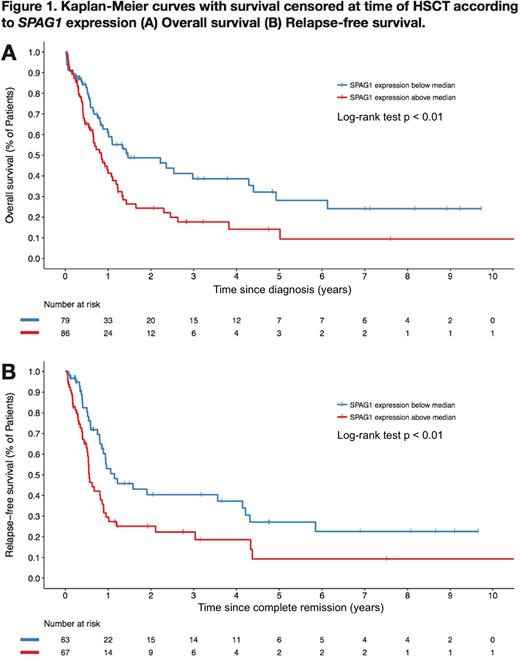
Results: We identified SPAG1 (Sperm Associated Antigen 1) as the gene with the highest HR for OS and RFS in the intermediate cytogenetic risk group. SPAG1 expression was dichotomized on the median RPKM value in the global cohort of 263 de novo AML specimens (RPKM cut-off 2.06). Using this cut-off, 79 patients had low expression of SPAG1 and 86 patients had high expression of SPAG1 in the intermediate cytogenetic risk cohort. Median age, sex, WBC at diagnosis and HSCT in CR1 rates were similar between the two groups. Patients with high expression of SPAG1 had a higher frequency of FLT3-ITD (51.2% vs 32.9%, p = 0.02) and DNMT3A mutations (51.2% vs 32.9%, p = 0.02). SPAG1-high patients were enriched in the NPM1/FLT3-ITD/DNMT3A triple positive mutation population (SPAG1-high 24/33 vs SPAG1-low 9/33, OR 3.00, p = 0.01). The frequencies of all other analyzed mutations were similar between both groups. CR rates did not differ between the two groups (SPAG1-high 77.9% vs SPAG1-low 79.7%, p = 0.77). In UVA with censorship at time of HSCT, SPAG1-high patients had worse OS (5-year estimates 14.2% vs 28.1%, HR 1.75, 95% CI 1.16-2.63, p < 0.01) and RFS (5-year estimates 9.3% vs 27.1%, HR 1.90, 95% CI 1.20-3.01, p < 0.01) (Figure 1). In MVA with censorship at time of HSCT, high expression of SPAG1 remained significantly associated with OS (HR 1.78, 95% CI 1.12-2.83, p = 0.01) and RFS (HR 2.34, 95% CI 1.38-3.96, p < 0.01). Furthermore, there was a significant interaction between SPAG1 GE and TD-HSCT (p = 0.09) in the RFS model. Importantly, SPAG1 had a lower prognostic impact for RFS in the model including TD-HSCT (HR 1.65, 95% CI 1.07-2.55, p = 0.02) compared with the model in which survival was censored at time of HSCT (HR 2.34, p < 0.01). High SPAG1 expression was also associated with worse OS in the TCGA AML dataset which is enriched in intermediate cytogenetic risk samples (p < 0.001).
Conclusion: In patients with intermediate cytogenetic risk AML, high expression of SPAG1 is independently associated with worse OS and RFS. The prognostic impact of SPAG1 expression on RFS is lower when adjusted for TD-HSCT indicating that the adverse prognosis conferred by high expression of this gene may be partially overcome by HSCT in CR1. Consequently, SPAG1 expression might help identify AML patients with intermediate cytogenetic risk who are most likely to benefit from HSCT in CR1. These results need to be validated in other independent cohorts and prospective studies before implementation into clinics.
Sauvageau:ExCellThera: Employment, Equity Ownership.
Author notes
Asterisk with author names denotes non-ASH members.